Nervous system models
A range of models have been developed and can be tailored to mimic specific aspects of the central and peripheral nervous systems. Some examples are provided here to give an overview of the technology, but more are described in our research publications. 3D hydrogels can provide a more realistic spatial environment for neurons and glial cells than traditional 2D culture systems. They allow continuous observation and controlled manipulation, thereby facilitating analysis of cellular interactions. They complement in vivo models which generally allow only snapshot views and offer limited scope for controlling complex variables.
If you would like to read about the use of advanced 3D cell culture systems including brain organoids, neurospheroids, and scaffold-based systems, which have been shown to recapitulate complex CNS microenvironments and processes then take a look at this book chapter: Onuwaje & Phillips 2020
Aligned tissue models of the central and peripheral nervous system
Nervous system tissues have a highly orientated structure which is recreated in our aligned tissue models. In tissues, endogenous forces are generated as resident cells attach to and remodel the extracellular matrix and this behaviour can be harnessed in collagen gels to allow strain generated by the cells to contract and align the substrate in a controllable, predictable and quantifiable manner.
Our peripheral nerve model recreates the key cellular and extracellular elements of peripheral nerve tissue. Neurons extend neurites within an aligned Type-I collagen matrix, guided and supported by Schwann cells that respond by forming myelin sheaths.
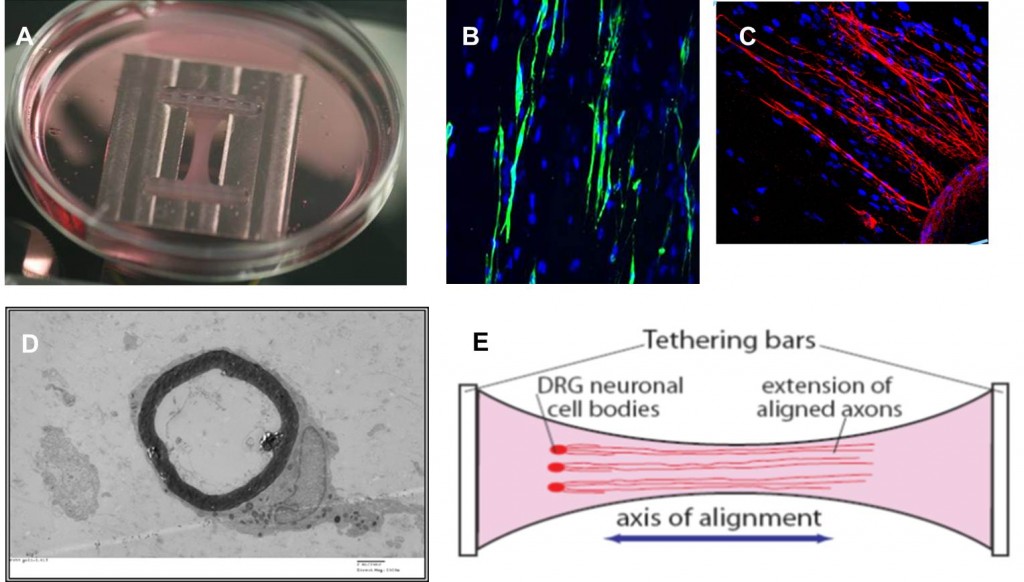
Peripheral nerve model: Schwann cells cultured in a tethered rectangular collagen gel (A) align with the direction of principal strain (B) and guide neuronal growth (C). Myelination follows (D), forming a model of the peripheral nerve (E).
This also provides a system for studying the behaviour of stem cell-derived cells with a view to therapeutic development. For example we have investigated the ability of differentiated human dental pulp-derived stem cells to myelinate neurons in vitro (Martens et al., 2013):
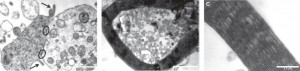
Differentiated human dental pulp stem cells myelinating neurons in aligned 3D co-culture (from Martens et al., 2013).
More recently, peripheral nerve models have been adapted for use in screening drugs that could be used to treat nerve injury (Rayner et al., 2018)
In our aligned CNS co-culture models, astrocytes or mixed CNS glia align within the 3D collagen environment. We have mapped the extent of astrocyte alignment in distinct parts of the gel, and this has been used to investigate the effect of astrocyte alignment on neuronal growth (East et al., 2010).
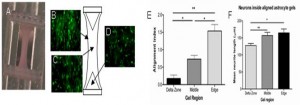
Astrocyte alignment model: Astrocytes in tethered collagen gels (A) contract and align in edge (B) and middle (C) zones, leaving unaligned cells in delta zones (D). Alignment was mapped in the three areas (E) and the system was used to assess the effect of astrocyte alignment on neuronal growth (F).
This approach has subsequently been miniaturised and adapted in collaboration with industry partners (O’Rourke et al., 2015) to provide robust and consistent 3D models for studying neurodegeneration (O’Rourke et al., 2017).
Astrocytes undergoing reactive gliosis
A great example of the importance to cells of a 3D environment is provided by our astrocyte models. In traditional monolayer cultures astrocytes exhibit a ‘reactive’ phenotype similar to that found in CNS astrocytes following injury. This makes the study of astrocytes with a physiological ‘resting’ phenotype (as found throughout the undamaged CNS) problematic in cell culture. We have shown that astrocytes in our 3D hydrogel system adopt a less reactive phenotype reminiscent of their counterparts in the undamaged CNS. When stimulated with a damage cue, they undergo reactive gliosis and mimic the cellular and molecular responses characteristic of CNS injury.
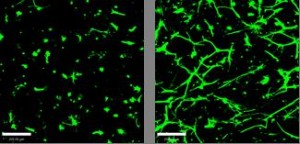
Astrocytes in 3D culture undergo reactive gliosis in response to simulated damage. After 15 days control cells resemble those found in normal undamaged tissue (left) whereas stimulated cells resemble those found in the glial scar (right)
These 3D astrocyte models are currently being used to screen cell therapies (e.g. stem cells) to assess the potential for implanted cells to cause reactive gliosis (see East et al., 2013).
Interface systems
Inhibitory interfaces surround sites of CNS injury, preventing neuronal regeneration. Modelling these cellular interfaces is useful for understanding the CNS response to damage and for developing new therapies to promote regeneration. We have developed 3D interface models that recreate key features of the response of CNS tissue to damage (such as spinal cord injury). We can simultaneously monitor glial and neuronal responses over time, so as the glial environment becomes more inhibitory we can measure the reduction in neuronal regeneration (East et al., 2012). Novel strategies for reducing inhibition and promoting regeneration can therefore be developed rationally in these systems.
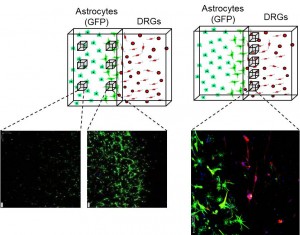
Interface model: Astrocytes become reactive at a 3D interface with dissociated DRGs, blocking neuronal regeneration and mimicking the inhibitory interface that forms at the border of a spinal cord injury lesion.